|
They travel in parallel lines through the empty tube. Since the electrons have a negative charge, they are repelled by the negative cathode and attracted to the positive anode. The increased random heat motion of the filament knocks electrons out of the surface of the filament, into the evacuated space of the tube. Modern vacuum tubes use thermionic emission, in which the cathode is made of a thin wire filament which is heated by a separate electric current passing through it. The positive ions were accelerated by the electric field toward the cathode, and when they collided with it they knocked electrons out of its surface these were the cathode rays. In the early experimental cold cathode vacuum tubes in which cathode rays were discovered, called Crookes tubes, this was done by using a high electrical potential of thousands of volts between the anode and the cathode to ionize the residual gas atoms in the tube. To release electrons into the tube, they first must be detached from the atoms of the cathode. The Maltese cross has no external electrical connection.Ĭathode rays are so named because they are emitted by the negative electrode, or cathode, in a vacuum tube. Cathode-ray tubes (CRTs) use a focused beam of electrons deflected by electric or magnetic fields to render an image on a screen.Ī diagram showing a Crookes tube connected to a high voltage supply. Thomson showed that cathode rays were composed of a previously unknown negatively charged particle, which was later named the electron. They were first observed in 1869 by German physicist Julius Plücker and Johann Wilhelm Hittorf, and were named in 1876 by Eugen Goldstein Kathodenstrahlen, or cathode rays. If an evacuated glass tube is equipped with two electrodes and a voltage is applied, glass behind the positive electrode is observed to glow, due to electrons emitted from the cathode (the electrode connected to the negative terminal of the voltage supply). Cathode rays are normally invisible in this demonstration with a Teltron tube, enough gas has been left in the tube that the gas atoms luminesce when struck by the fast-moving electrons.Ĭathode rays ( electron beam or e-beam) are streams of electrons observed in discharge tubes. This phosphorescence gets stronger as more gas is pumped out but the lines in the gas get fainter.A beam of cathode rays in a vacuum tube bent into a circle by a magnetic field generated by a Helmholtz coil. These get their name because they come from the (conventionally) negative of the two terminals which lead the current in and out, which Whewell, then Master of Trinity College, Cambridge, called ‘cathode’ after Faraday had asked him for suggestions.* They appear as straight lines diverging from points on the cathode and show up as phosphorescence when they reach the glass walls. Soon after the stage when the density allows the discharge to pass most easily the brilliantly coloured display of lights in the tube begins to show a new feature, called the cathode rays. Some gas is needed, in fact, but not too much. 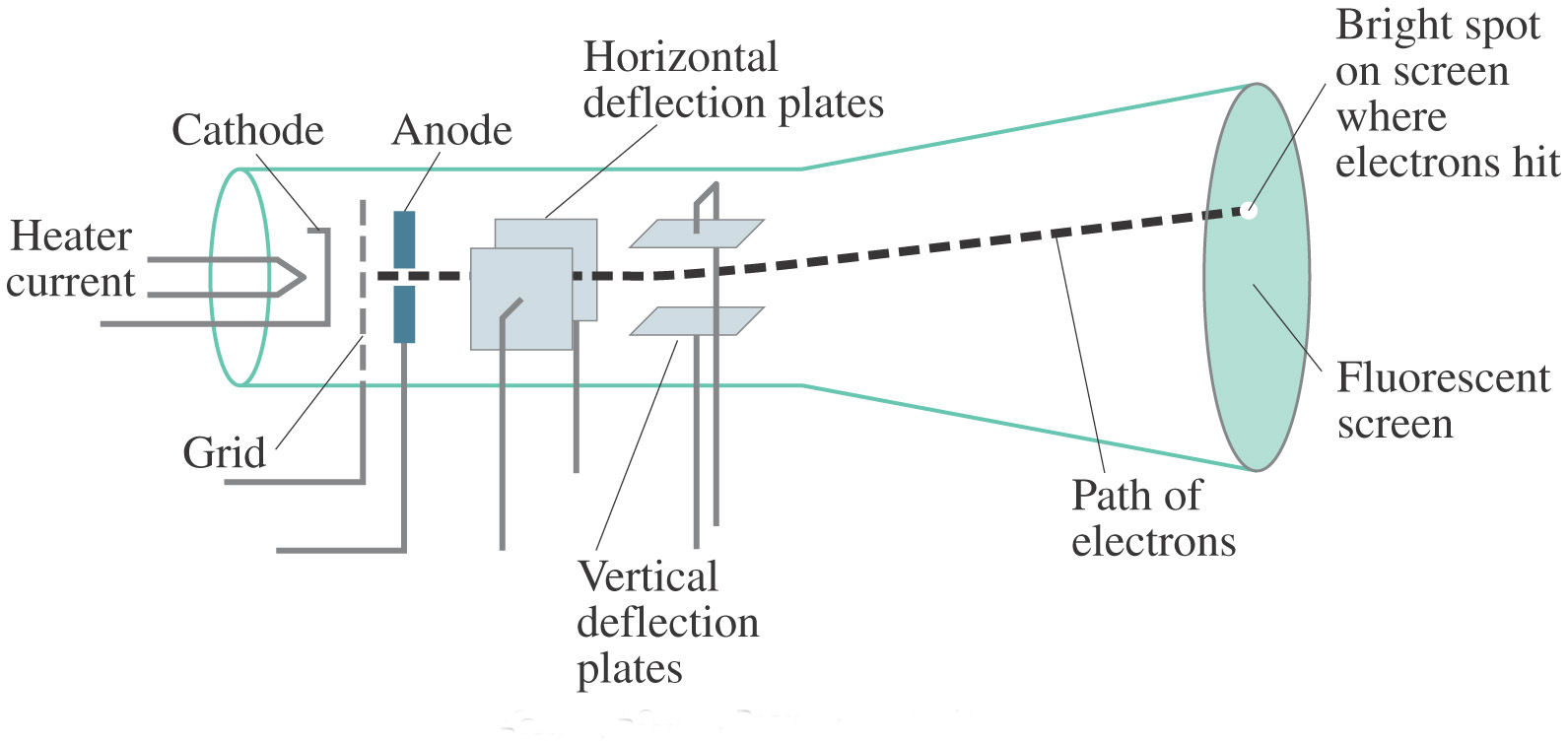
When for example the air in a glass tube is reduced to a density about 1/ 3000 of the normal an electric current can be passed through it with far less than the voltage which would be needed if most of the air had not been pumped out, though if the density is reduced by still another factor of io or so it again becomes harder to pass a current. 
These are one of the many strange phenomena which occur when electricity is made to pass through highly rarificd gases. The experiment in question had nothing to do with these waves, it concerned the nature of cathode rays. Hertz, the first man to prove experimentally that electromagnetic waves exist and can be propagated in free space. In making sense of a complicated set of scientific facts, or claimed facts, it often happens that one turns out to be wrong, but it is rare for the villain to be an experiment by as great a physicist as H.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
